Now, the Ocean Acidification is a lot of noise about a small potential. But to answer it properly, you have to realize a lot of past history and the normal effects of the oceans.
First, let's look at the claims from the TED talk:
"But I'll tell you as more carbon dioxide enters the ocean, the seawater pH goes down. And this basically means that there is an increase in ocean acidity."There has already been an increase in ocean acidity of 26 percent since pre-industrial times, which is directly due to human activities. Unless we can start slowing down our carbon dioxide emissions, we're expecting an increase in ocean acidity of 170 percent by the end of this century.Lets start with the agreements:They were placed in seawater at a pH we're expecting by the year 2100. After six months, the coral has almost completely dissolved.
ph is dropping: The water is becoming more neutral. Since the Ocean starts as a light base, it still has a ways to go till it is neutral and even further still till it becomes acidic. The slime on fish is to protect them from a ocean that is not acidic, but actually a light base.
The 26 percent increase since industrial times: This is again where we run into a data issue, and an argument. In the report she uses the graph from is from Dr. Richard A. Feely, and Feely’s four-page report:The problem with that was, well:Carbon Dioxide and Our Ocean Legacy.This chart, titled “Historical & Projected pH & Dissolved Co2,” begins at 1850. Feely testified before Congress in 2010—using the same data that shows a decline in seawater pH (making it more acidic) that appears to coincide with increasing atmospheric carbon dioxide.So, that chart has a huge omission of data. And the process with which they needed to get it? Well, the authors wouldn't turn it over for review, so in the end it required a FOIA request to get it.Mike Wallace is a hydrologist with nearly 30 years’ experience, who is now working on his Ph.D. in nanogeosciences at the University of New Mexico. In the course of his studies, he uncovered a startling data omission that he told me: “eclipses even the so-called climategate event.” Feely’s work is based on computer models that don’t line up with real-world data—which Feely acknowledged in email communications with Wallace (which I have read). And, as Wallace determined, there is real world data. Feely, and his coauthor Dr. Christopher L. Sabine, PMEL Director, omitted 80 years of data, which incorporate more than 2 million records of ocean pH levels.
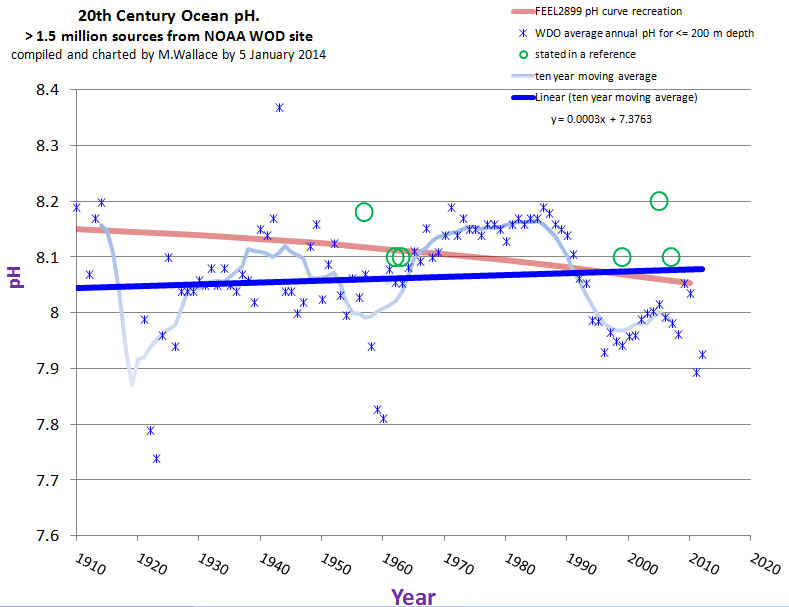
The interesting bit was the resulting data was obtained by Wallace, from NOAA with the missing 1.5 million points, and put into a chart:
This is rather troubling. With all of the data inserted, not modeled by Feely, the oceans show a slight, to use the vernacular, de-acidification, though it is easy to see a drop in ph in the last 20 years. So, some of the basic assumptions from the talk can be rejected. That being said, what was found after that?
Let's ignore the long, long term trend and look from about 1970 onward, where we have better data, and a definitive change in CO2 levels in the ocean. This does show a trend towards neutral. Of about .018 +/- .001 per decade. This might result in a change of .2 ph in 100 years. So, a small change, if the ph is changed simply because of co2 in the atmosphere. There is Henry's Law also, but I will include that later.
Conclusion: pH is dropping. Not as fast as Freely said. So the projections are extreme. But that doesn't mean they are not changing, and skeptics agree that there is the potential for a change in pH.
_______________________________
But this is where it is important to make it relative.
The ocean is not one level ph. Nor even really well blended.
We can see a range of ph, and even within one location, ph can vary a large amount:
So the rate of change in ph is less than the amount of error in many locations, and less than the cycle of ph that these areas are exposed to.
What about the coral and shell dissolving? Well, when they tried to recreate that effect out in the oceans, instead of in a tank, the results were far less. The experiment was interesting:These observations reveal a continuum of month-long pH variability with standard deviations from 0.004 to 0.277 and ranges spanning 0.024 to 1.430 pH units. The nature of the observed variability was also highly site-dependent, with characteristic diel, semi-diurnal, and stochastic patterns of varying amplitudes. These biome-specific pH signatures disclose current levels of exposure to both high and low dissolved CO2, often demonstrating that resident organisms are already experiencing pH regimes that are not predicted until 2100.
This really was a cool study. https://www.nature.com/articles/srep00413.pdfGeorgiou, et al. did their experiments in situ the open ocean by building enclosures that are open on two sides and on the bottom and raising the level of CO2 in the “treated” enclosures. They compared the results to nearby corals that were in enclosures with no added CO2 (the “controls”). They found no reef growth differences between the two environments. This implies a high degree of tolerance to ocean acidification. The enclosures for injecting CO2 and the control enclosures are called FOCE (Free Ocean Carbon Enrichment) and are described in Kline, et al., 2012. They describe a very ingenious way to measure the effects of sea-water carbon dioxide concentration differences on corals in-situ.
They simulated 600-1000ppm CO2 levels.
Conclusion: pH level change quite a bit in The effect on coral will be less than expected, when tested in real world situations, which is encouraging.
__________________________________________________
Henry's Law and the rest:
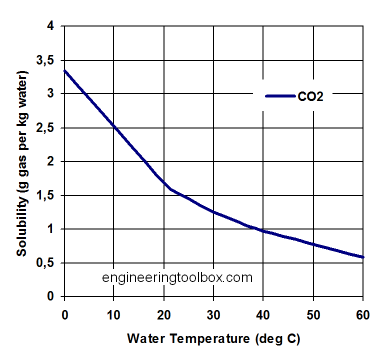
Henry's law is the beer law. It states how much CO2 or otherwise can be in a liquid at what pressure and temperature.
CO2 is trapped in the ocean. In fact, it is an interesting bit that the oceans hold 38,000Gt of CO2, and we put out, like 9 or 10.
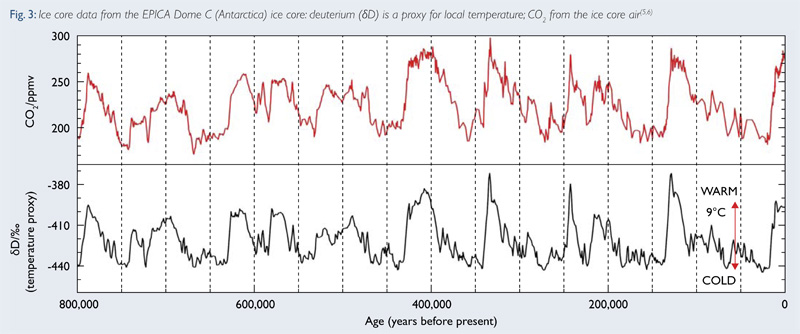
If the oceans warm up, they can't hold as much CO2. As the cool, they absorb more. Looking at the Volstok Ice samples, we find that CO2 falls behind the warming of the other interglacial periods. By about 600 years. What happens is, as the Earth warms the oceans warm, but slower than the atmosphere, and more CO2 is released. Then as the air cools, then the oceans cool and it absorbs more of the CO2.
That is why you had a net rise of CO2 following 1850 till 1950 - that was natural in origin, following the warming of the ocean. Henry's law is why. Humans, in the post WWII development finally started to put out CO2 in a measurable amount.
The atmosphere? Well, that has only 843Gt of CO2. I am not saying all of the CO2 is from a warming ocean - but a large portion is, and the natural cycles drive a large portion of the CO2.The ocean surface area is 360 million sq. kilometers, or 360 trillion sq. meters. The top meter is 360 trillion tons of water. A change of temperature of one degree for cold water changes the solubility by:
360 X 1012 tons = 360 X 1015 kg X 0.08 g/kg = 28.8 X 1015 g CO2 or 28.8 Petagrams CO2. A tenth of a degree temperature change changes solubility by 2.88 Petagrams CO2. This is about 780 Gigatons carbon equivalent. (3.7 grams CO2 = 1 gram carbon.)
SO, what does this mean? Well, go back 10,000 years, and the oceans were a lot cooler than now - we are normally in an Ice Age. So the Co2 levels are higher - and hence, the pH would be far more acidic. Well, more neutral.
And we still find large deposits of shells. It actually is one of the proxy items used to find out past temperature.
Conclusion: CO2 levels in the ocean cycle up and down, and the corals and related animals survived those period also.
________________________________________
Over all conclusion from the skeptic side:
The ocean data shows less change in pH than the Feely reports, when all data is accounted for.
Projections of damage from the Feely report would not likely arise because of the problem with the conclusions in that report.
Actual in ocean testing of the extremes from the Feely report show no adverse effect.
Oceans have cycled at higher pH extremes than proposed in the Feely report, and ocean life still has survived, including corals.
I could expound a bit, there is quite a bit on the subject, but as a quick response the pH levels projected from Feely's paper should not be taken as truth, and testing in the ocean has shown no damage to coral. pH should change to a more neutral position, but it also should be expected to fall within the normal margin of error that ocean dwelling creatures are used to in the next 100 years.
Oh, and if you want to be taken seriously as a scientist, you should allow your work to be examined. Feely didn't let other people try and replicate the work, resulting in a FOIA filing. The resulted in finding 80 years of data was removed to make the claims he did.
That should be a huge warning sign to any body.